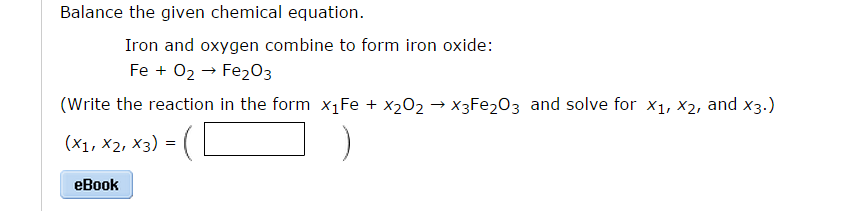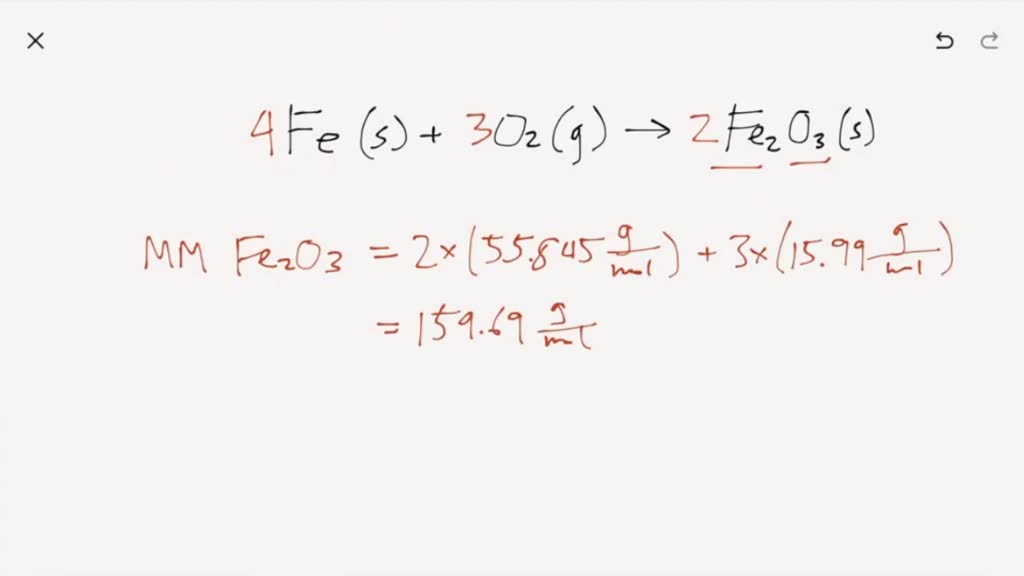
SOLVED:Iron can react with \mathrm{O}_{2} to produce two different oxides, \mathrm{Fe}_{2} \mathrm{O}_{3}(s) or \mathrm{Fe}_{3} \mathrm{O}_{4}(s) . Write the chemical equations that describe both reactions. If 167.6 grams of Fe react completely with

Causes chemistry of rusting rust prevention introduction to oxidation reduction REDOX reactions gcse igcse KS4 science chemistry revision notes revising

What does it mean for an equation to be balanced? We have the same number of each type of atom on each side of the equation. Make sure you have balanced. -

Chapter 8.1. Warm up 1. What is a chemical reaction? 2. What is conservation of mass? 3. How are the two concepts above related? 4. When heated, solid. - ppt download








:max_bytes(150000):strip_icc()/BalanceEquations1-56a132765f9b58b7d0bcf535.png)